All published articles of this journal are available on ScienceDirect.
Cardiac Arrhythmias Following Left Ventricular Aneurysm Resection: A 10-Year Retrospective Study
Abstract
Introduction
Left Ventricular Aneurysm (LVA) is a serious complication of myocardial infarction that can lead to impaired ventricular function and life-threatening ventricular arrhythmias. Data on perioperative arrhythmic patterns and associated clinical factors following LVA resection, particularly with long-term follow-up, remain limited. We aimed to characterize perioperative arrhythmias and their clinical correlates following surgical LVA resection with mid-term follow-up.
Materials and Methods
Retrospective cross-sectional study with longitudinal ECG and echocardiographic follow-up, 29 patients who underwent surgical LVA resection between 2011 and 2022 at a tertiary cardiac center were evaluated. Demographic characteristics, comorbidities, lipid status, perioperative complications, Electrocardiographic (ECG) findings, echocardiographic parameters, length of hospital stay, and mortality were extracted from medical records and follow-up assessments. ECGs were analyzed preoperatively, during Intensive Care Unit (ICU) admission, at ward admission, and at 6 months and 2 years postoperatively. Left Ventricular Ejection Fraction (LVEF) was assessed longitudinally.
Results
Preoperative ventricular arrhythmias were observed in 34.5% of patients, while atrial fibrillation was present in 17.2%, with no significant sex-based differences. Postoperative arrhythmias occurred in 20.7% of patients. While 72.4% of patients had normal ECGs at ICU admission, observed abnormalities were significantly more frequent among male patients (p = 0.040). Dyslipidemia was associated in univariable analysis with abnormal postoperative ECG findings during ICU and ward admission (p = 0.012 and p = 0.047, respectively). Mean LVEF improved significantly from 33.4% ± 8.1% preoperatively to 42.9% ± 6.9% at 2-year follow-up (p < 0.05). Six patients died during the early postoperative period, primarily due to low cardiac output syndrome and cardiac tamponade; two additional deaths were identified during follow-up, with undetermined causes.
Discussion
Surgical resection of LVA is associated with significant improvement in left ventricular function; however, cardiac arrhythmias remain relatively common in the perioperative period. Male sex and dyslipidemia appear to be associated with a higher burden of postoperative ECG abnormalities.
Conclusion
These findings underscore the importance of meticulous perioperative monitoring and risk-factor optimization, and closer monitoring of cardiovascular risk factors may be warranted following LVA resection.
1. INTRODUCTION
Left Ventricular Aneurysm (LVA) is a well-recognized consequence of myocardial infarction that can lead to life-threatening sequelae, such as cardiac arrhythmia, cardiac rupture, and heart failure [1]. A recent review by Jan et al. reported that patients with apical aneurysms had an overall mortality rate of approximately 9%-10.5% over a follow-up period of 2 to 6.5 years [2]. Following a Myocardial Infarction (MI) and the replacement of the ventricular wall with fibrous tissue, the weakened wall tends to develop a bulged area on the ventricular wall, a dyskinetic outpouching of the left ventricular wall, and contributes to diminished left ventricular ejection fraction [3, 4]. The reported incidence of LVA following Myocardial Infarction (MI) ranges from 10% to 35%. However, it has declined to approximately 5% to 15%, due to the benefits of rapid reperfusion therapy and medications that prevent ventricular remodeling [5, 6]. Surgical resection of the aneurysm and reconstruction of the myocardial wall are performed to improve hemodynamics and to improve LV geometry and function [4]. However, despite the benefits of LVA repair, postoperative cardiac arrhythmias remain a significant concern, impacting morbidity and mortality. Cardiac arrhythmias following LVA resection result from multiple pathophysiological mechanisms. The development of malignant ventricular pathways, particularly at the interface between viable and fibrotic myocardium [7].
Myocardial fibrosis and scarring, which are inherent to both the infarction and surgical intervention, can create substrates for reentrant arrhythmias [8]. Moreover, surgical manipulations and direct mechanical irritation of the myocardium and pericardium are among the postoperative arrhythmias after cardiothoracic surgery. Increased sympathetic and hormonal activity, systemic inflammatory response, and complement pathway activation may also contribute to electrical instability [9]. Among post-surgical arrhythmias, ventricular arrhythmias, including Ventricular Fibrillation (VF) and Ventricular Tachycardia (VT), have the highest risk of sudden cardiac death [10, 11]. They often develop from reentry cycles in scarred tissue of infarcted myocardium and require immediate management. The treatment encompasses antiarrhythmic therapy, catheter ablation, or Implantable Cardioverter-Defibrillator (ICD) placement [10, 12]. Supraventricular arrhythmias, such as Atrial Fibrillation (AF), are also prevalent following LVA resection, particularly in patients with preexisting atrial enlargement or structural heart disease [13]. AF can lead to hemodynamic deterioration and an increased risk of thromboembolism, necessitating the implementation of anticoagulation and rhythm- or rate-control strategies [14]. Despite numerous studies focusing on arrhythmias after LVA resection, notable gaps persist in the existing literature. While previous research has investigated treatment outcomes of LVA resection and the prognosis of arrhythmias, few studies have examined patient-specific risk factors, such as sex-related differences and metabolic influences, including lipid profiles [15, 16]. Additionally, to the best of our knowledge, long-term post-surgical follow-up of patients with LVA resection has not yet been studied. Therefore, this study aimed to investigate the incidence of cardiac arrhythmias following LVA resection and to identify their clinical and perioperative correlates.
2. MATERIALS AND METHODS
2.1. Participants and Study Design
This retrospective cross-sectional study was conducted at the cardiac surgery ward of Imam Reza Hospital, Mashhad, Iran, in 2024. Participants were recruited if they had been diagnosed with an LVA following a previous MI and had undergone aneurysm resection surgery between February 2011 and January 2022. Data were primarily obtained from patients’ hospital records, with confidentiality maintained through anonymized profiles. Accordingly, the ethics committee waived the need for informed consent for the use of retrospective data. However, patients lacking two-year postoperative echocardiographic data were contacted directly. The study protocol and follow-up procedures were explained, and informed consent was obtained from those willing to participate and undergo the assessment.
Exclusion criteria included: (1) patients with incomplete data who declined further participation, (2) congenital or post-traumatic LVAs, (3) concomitant LVA with significant valvular disease, (4) a history of stroke, (5) renal failure, or (6) arrhythmias associated with electrolyte imbalances.
The present study did not include a non-surgical control group. Due to ethical and clinical considerations, withholding surgical treatment from symptomatic patients with LVA and impaired ventricular function was not feasible. Therefore, the primary aim of this study was to describe the incidence of postoperative arrhythmias and identify associated clinical factors, rather than establish causality.
2.2. Data Collection and Diagnostic Criteria
Demographic and clinical data, including age, sex, Body Mass Index (BMI), intraoperative and postoperative complications, duration of hospital stay, relevant medical history (such as hypertension, dyslipidemia, diabetes mellitus, pulmonary disease, and prior myocardial infarction), and mortality rates, were obtained from the Hospital Information System (HIS) and archived hospital records.
Preoperative angiography and transthoracic or transesophageal echocardiography had also been performed in all patients to assess left ventricular dimensions, function, and valvular status, and the results were documented in their hospital records. The diagnosis of LVA had been initially established based on angiographic evidence of paradoxical wall motion and subsequently confirmed during surgery. Angiographic diagnosis was made according to the CASS criteria, which define an LVA as a segment of the left ventricular wall that protrudes from the normal contour and exhibits either akinesis or dyskinesis.
2.3. Preoperative Care and Surgical Procedure
All patients received standard preoperative care, including heart failure treatment protocols such as beta-blockers, ACE inhibitors or ARBs, diuretics, statins, and others when indicated. Electrolyte levels and volume status were corrected before surgery, and preoperative angiography and echocardiography were performed to assess ventricular function, aneurysm details, and valvular condition.
All patients had undergone LVA resection via median sternotomy under cardiopulmonary bypass. Aneurysmectomy had been performed with excision of the fibrotic segment, followed by ventricular reconstruction using either linear closure or endoventricular circular patch plasty (Dor technique), depending on aneurysm size, location, and residual ventricular geometry. Concomitant Coronary Artery Bypass Grafting (CABG) had been performed when indicated.
2.4. Follow-up and Outcome Assessment
Electrocardiography (ECG) had been routinely performed preoperatively. Postoperative continuous ECG monitoring was also performed immediately after surgery, in the Intensive Care Unit (ICU) upon admission, and during the ward stay to assess arrhythmias.
Transthoracic echocardiography had also been recorded at four time points: 1 week before surgery, immediately after aneurysm resection, 6 months post-surgery, and 2 years post-surgery. A detailed checklist was developed, and all data were collected by two board-certified cardiac surgeons and an experienced nurse.
Primary outcomes included incidence and types of cardiac arrhythmias detected on ECG across perioperative and follow-up periods. Secondary outcomes comprised changes in LVEF over time, intraoperative/postoperative complications, hospital length of stay, and mortality.
2.5. Statistical Analysis
Statistical analyses were performed using SPSS version 25 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± Standard Deviation (SD) or median with Interquartile Range (IQR), while categorical variables were reported as frequencies and percentages. The normality of data distribution was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests. For between-group comparisons, Mann-Whitney U and Wilcoxon tests (for non-normal data) were used for continuous variables with non-normal distribution, and the repeated-measured ANOVA test was used for normally distributed continuous ones. Moreover, chi-square and Fisher's exact tests were applied for categorical variables. Changes in LVEF over time were assessed using repeated measures ANOVA. The normality of LVEF at each time point was verified using the Shapiro-Wilk test. The assumption of sphericity was assessed using Mauchly’s test, and when sphericity was violated, the Greenhouse-Geisser correction was applied to adjust the degrees of freedom. A p-value of <0.05 was considered statistically significant. Post-hoc pairwise comparisons were not conducted due to the small sample size and the exploratory nature of the analysis. Additionally, multivariate analyses, such as logistic regression, were not performed because the limited sample size would have rendered such models underpowered and potentially unstable.
3. RESULTS
3.1.Patient Demographics and Preoperative Characteristics
A total of 29 patients were enrolled in the study, including 19 males (65.5%) and 10 females (34.5%), with a mean age of 56.6 ± 17.9 years. Demographic characteristics, past medical history, and substance use history are summarized in Table 1. Myocardial infarction (22 patients, 75.9%) and hypertension (10 patients, 34.5%) were the most prevalent comorbidities. The anterior type was the most common left ventricular aneurysm, observed in 79.3% of the study population. It is also evident that older age groups constitute the majority of the sample, with the 60-69 and 70-79 age groups comprising 58.6% of the total population. Detailed demographic characteristics and prior medical history of the participants are presented in (Table 1).
Table 2 summarizes preoperative and postoperative ECG findings and their association with gender. Preoperatively, ventricular arrhythmias were identified in 10 patients (34.48%), including Premature Ventricular Contractions (PVCs) in six patients and Ventricular Tachycardia (VT) in four patients. Atrial Fibrillation (AF) was present in five patients (17.24%). No significant sex-related differences were observed in preoperative ECG findings (p = 0.765).
Postoperative arrhythmias occurred in six patients (20.7%), with a comparable incidence between females and males (20.0% vs. 21.1%, p < 0.999). At ICU admission following surgery, most patients (72.4%) demonstrated normal ECG findings; however, ECG abnormalities were more frequent among male patients, resulting in a statistically significant difference between the sexes (p = 0.040). Two patients died before ICU ECG assessment (cardiac tamponade and low cardiac output syndrome).
| Characteristic | Category | Number (%) |
|---|---|---|
| Sex | Male | 19 (65.5) |
| Female | 10 (34.5) | |
| Age group (years) | <20 | 1 (3.4) |
| 20–29 | 3 (10.3) | |
| 30–39 | 1 (3.4) | |
| 40–49 | 2 (6.9) | |
| 50–59 | 5 (17.2) | |
| 60–69 | 10 (34.5) | |
| 70–79 | 7 (24.1) | |
| Comorbidities | Hypertension | 10 (34.5) |
| Dyslipidemia | 8 (27.6) | |
| Diabetes mellitus | 13 (44.8) | |
| Congenital heart disease | 0 (0) | |
| Pulmonary disease | 4 (13.8) | |
| Renal disease | 2 (6.9) | |
| Prior angioplasty | 20 (69.0) | |
| Cerebrovascular disease | 0 (0) | |
| Prior myocardial infarction | 22 (75.9) | |
| Aneurysm location | Anterior | 23 (79.3) |
| Posterior | 2 (6.9) | |
| Inferior | 4 (13.8) | |
| Substance use history | Present | 10 (34.5) |
| Absent | 19 (65.5) |
| ECG Timing | ECG Finding | Female, n (%) | Male, n (%) | P-value |
|---|---|---|---|---|
| Postoperative arrhythmia | Present | 2 (20.0) | 4 (21.1) | 0.999 |
| Absent | 8 (80.0) | 15 (78.9) | ||
| Preoperative ECG | Normal | 4 (40) | 9 (47.4) | 0.765 |
| VT | 2 (20) | 2 (10.5) | ||
| LBBB | 0 (0) | 2 (10.5) | ||
| AF | 2 (20) | 2 (10.5) | ||
| PVC | 2 (20) | 3 (15.8) | ||
| AF + PVC | 0 (0) | 1 (5.3) | ||
| ICU ECG (postoperative) | Normal | 6 (60.0) | 15 (78.9) | 0.040 |
| Deceased | 2 (20.0) | 0 (0) | ||
| AF + PVC | 0 (0) | 2 (10.5) | ||
| VT + PVC | 0 (0) | 2 (10.5) | ||
| PVC | 0 (0) | 2 (20) | ||
| Ward ECG (postoperative) | Normal | 6 (60.0) | 15 (78.9) | 0.127 |
| Deceased | 4 (40.0) | 2 (10.5) | ||
| AF + PVC | 0 (0) | 2 (10.5) |
Data were analyzed using the chi-square or Fisher’s exact test.
ECG assessment during ward admission showed normal findings in 72.4% of patients, with no significant sex-based differences (p = 0.127). Combined AF and PVC was the most frequently observed postoperative arrhythmia at this stage. By the time of ward evaluation, six patients had died.
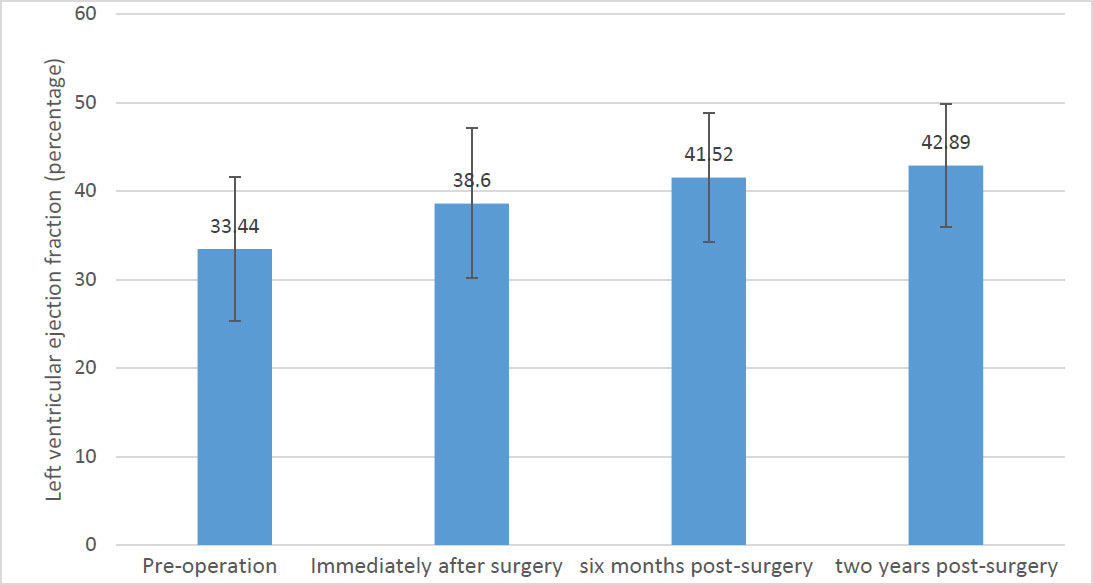
Figure 1 shows changes in LVEF across the predefined time points. The mean LVEF was 33.44 ± 8.13% preoperatively and increased to 42.89 ± 6.90% at 2 years postoperatively. Repeated-measures ANOVA revealed a statistically significant difference in LVEF across the assessed time points (p < 0.05).
Table 3 presents the relationship between patients' history of dyslipidemia and the occurrence of arrhythmias. Preoperative ECG analysis revealed no significant difference between dyslipidemia and normal Vs abnormal ECG findings (P=0.238). However, postoperatively, a higher proportion of patients without dyslipidemia maintained normal ECG readings in the ICU (81%) compared to those with dyslipidemia (50%) (p=0.012). This trend continued in the ward, where 81% of patients without dyslipidemia had normal ECGs, compared with 50% of those with dyslipidemia (p=0.047). Statistical analysis using the chi-square test and Fisher's exact test confirmed these findings.
| ECG Timing | ECG Finding | No Dyslipidemia, n (%) | Dyslipidemia, n (%) | P-value | |
|---|---|---|---|---|---|
| Postoperative arrhythmia | Present | 4 (19.0) | 2 (25.0) | 0.999 | |
| Absent | 17 (81.0) | 6 (75.0) | |||
| Preoperative ECG | Normal | 11 (57.1) | 2 (28.6) | 0.238 | |
| Abnormal ECG |
LBBB | 0 (0) | 2 (28.6) | ||
| VT | 2 (9.5) | 2 (25.0) | |||
| AF | 3 (19.0) | 1 (14.3) | |||
| PVC | 5 (23.8) | 0 (0) | |||
| AF + PVC | 0 (0) | 1 (28.6) | |||
| ICU ECG (postoperative) | Normal | 17 (81.0) | 4 (50.0) | 0.012 | |
| Deceased | 0 (0) | 2 (25.0) | |||
| AF + PVC | 0 (0) | 2 (25.0) | |||
| VT + PVC | 2 (9.5) | 0 (0) | |||
| PVC | 2 (9.5) | 0 (0) | |||
| Ward ECG (postoperative) | Normal | 17 (81.0) | 4 (50.0) | 0.047 | |
| Deceased | 4 (19.0) | 2 (25.0) | |||
| AF + PVC | 0 (0) | 2 (25.0) | |||
Data were analyzed using the Chi-square or Fisher’s exact test.
| Clinical Variable | Status | Length of Hospital Stay (days), Median (IQR) | P-value | |
|---|---|---|---|---|
| Preoperative arrhythmia type | Ventricular arrhythmia | Present | 20 (4.5–141.5) | 0.171* |
| Absent | 12 (7–17.5) | |||
| Atrial arrhythmia | VT | 13.5 (13–14) | 0.928** | |
| PVC | 16.5 (3–30) | |||
| None | 12 (7–21) | |||
| Postoperative arrhythmia | Present | 14 (3–30) | 0.787* | |
| Absent | 12 (7–21) | |||
The association between surgical complications, cardiac arrhythmias, and hospital admission duration is presented in Table 4. Patients with atrial arrhythmia, VT, or PVC were admitted longer than those without any arrhythmia; however, no significant association was found between cardiac arrhythmias and hospital admission duration.
In addition, the association between cardiac arrhythmias and patients’ age was assessed using the Chi-square test, and no significant association was observed (P>0.05). Moreover, no association was found between preoperative pulmonary disease and any type of cardiac arrhythmia (P<0.05).
3.2. Mortality
During the early postoperative period, mortality occurred in six patients (20.7%), either during the ICU stay or upon admission to the cardiac surgery ward. Four deaths were attributed to low cardiac output syndrome, and two resulted from cardiac tamponade. By the time the study commenced, the total number of deceased patients had reached eight; however, the causes of death in the remaining two cases could not be determined based on telephone follow-up.
4. DISCUSSION
This cross-sectional study offers insights into the incidence, characteristics, and outcomes of cardiac arrhythmias following LVA resection, a procedure performed to restore ventricular geometry and subsequently improve cardiac function. The results confirm that ventricular arrhythmias, particularly PVCs, are a significant postoperative complication, affecting 20.7% of the study's cohort. One of the most notable findings is the high incidence of PVCs in both preoperative and postoperative stages, suggesting a possible persistent electrophysiological abnormality following myocardial infarction and subsequent LVA. The study's findings can be explained by several factors. First, the pathophysiology of arrhythmias post-surgery is multifactorial. LVAs are often the result of prior myocardial infarction, which leads to myocardial scarring and fibrosis [17]. The fibrotic architecture of the aneurysm acts as a substrate for electrical instability, primarily by altering myocardial conduction properties and fostering reentrant circuits [8, 17, 18]. The resection of LVA may mitigate some of these abnormalities, as evidenced by the significant improvement in LVEF from a mean of 33.4% preoperatively to 42.9% at two years post-surgery. This suggests that while surgery improves overall cardiac function, it does not entirely resolve the arrhythmic risk, likely due to the presence of scar tissue and impaired electrical conduction in the remaining myocardium [12, 19-21]. The pathogenesis of arrhythmias following LVA resection reflects an interaction between a pre-existing scar-based substrate and acute perioperative triggers, consistent with contemporary concepts of scar-related ventricular arrhythmogenesis and postoperative rhythm vulnerability [22, 23]. Although aneurysmectomy removes dyskinetic scar, the peri-scar/border-zone where surviving myocyte strands are interspersed with fibrosis often remains a critical site for slow, heterogeneous conduction and re-entrant VT circuits [22, 24]. Experimental mapping in healed MI demonstrates that the infarct scar border supports VT circuits, with conduction impairment and spatial electrophysiologic heterogeneity playing a key mechanistic role [25]. More broadly, electrical remodeling and conduction slowing in structurally remodeled myocardium are well-recognized prerequisites for reentry and complex ventricular ectopy [25]. Additionally, the occurrence of arrhythmias post-surgery could be partly due to surgical trauma and ischemia-reperfusion injury, both of which contribute to electrical disturbances in the heart [23, 26]. The early postoperative period (ICU phase) is a high-risk window during which acute triggers act upon the underlying substrate. Post-cardiac surgery reviews identify multiple contributors, including hemodynamic stress, metabolic abnormalities, electrolyte disturbances (notably K+/Mg2+ shifts), and increased adrenergic tone (including stress response and inotropic agents) [23, 27].
Regarding mechanical factors, sudden changes in loading conditions can acutely influence electrophysiology via mechano-electric feedback; direct intraoperative human data demonstrate that abrupt increases in ventricular loading can measurably alter electrophysiologic parameters, providing physiologic support for “mechanical stress” as a plausible trigger on a vulnerable substrate [28]. Contemporary reviews also emphasize “electromechanical reciprocity” as a clinically relevant contributor to arrhythmogenesis in susceptible hearts [29]. One of the most consistent findings in our study was the significant improvement in LVEF after surgery. The preoperative LVEF was 33.4% ± 8.1%, and it increased to 42.9% ± 6.9% two years postoperatively. Sartipy et al. (2008) examined the prevalence of arrhythmias in patients with both ventricular aneurysms and ventricular tachycardia following surgery. In their study of 63 patients, they observed a decrease in arrhythmia rates post-surgery, with a two-year survival rate of approximately 85% and a five-year survival rate of 62%. The leading cause of death was severe heart failure. Our findings align closely with those of Sartipy et al., as we also observed a reduction in pathological ECG changes post-surgery. Additionally, our two-year survival rate was approximately 80%, consistent with their results [30]. Another finding of interest is the increased mortality rate among patients who developed postoperative complications. A total of six patients (20.7%) died after surgery, with the majority of deaths attributed to low cardiac output syndrome (4 patients) and cardiac tamponade (2 patients). The role of low cardiac output syndrome in post-surgical mortality is well-documented, often resulting from inadequate myocardial function following the removal of an aneurysmal segment [31]. The observation that tamponade, a condition in which fluid accumulates in the pericardial sac and restricts normal cardiac filling and reduces cardiac output, was also a significant contributor to mortality, and it highlights the need for close postoperative monitoring, particularly in patients with impaired ventricular function [32, 33].
Our data showed a significant correlation between dyslipidemia and pathological postoperative ECG changes, which is another critical aspect of the study, consistent with previous studies [34]. Liu et al.'s (2006) study, examining eight patients with acute MI, revealed that higher levels of low-density Lipoprotein Cholesterol (LDL-C) were significantly associated with an increased risk of developing VT or VF during the acute stage of MI [34]. Mechanistically, changes in membrane lipid composition (including cholesterol content) can influence membrane protein conformation and function, with downstream effects on cardiac excitability. In near-native membrane experiments, cholesterol depletion significantly reduced Na+/K+-ATPase activity and altered reaction kinetics, supporting the principle that membrane lipid environment can modulate key electrogenic transporters relevant to resting membrane potential stability and ectopy thresholds [35]. In our study, patients with a history of dyslipidemia were more likely to exhibit abnormal postoperative ECGs, as well as more severe arrhythmias such as AF and PVCs. This finding suggests that dyslipidemia may contribute to the underlying pathophysiology of arrhythmias, possibly through its effects on myocardial ischemia, endothelial dysfunction, and the promotion of fibrosis [36, 37]. The increased vulnerability to arrhythmias in these patients could be related to a combination of factors, including the accelerated progression of coronary artery disease and the resulting impact on the electrical stability of the myocardium. Interestingly, no significant statistical correlation was found between specific arrhythmia types and age groups in our cohort. This apparent lack of an age-related effect may be attributable to the relatively small sample size, which likely limited the statistical power to detect subtle differences. However, the broader literature suggests that the incidence of arrhythmias generally increases with age [38, 39]. In terms of comorbidities, our findings indicated no significant association between preoperative pulmonary disease and arrhythmias (p > 0.05). This contrasts with the study by Serrano Jr. et al. (2010) [40], which demonstrated that aneurysmectomy in patients with severe left ventricular dysfunction was associated with a higher rate of pulmonary tract infections (73% in patients who expired). The discrepancy between our findings and those of Serrano Jr. et al. (2010) [40] may be attributed to our smaller sample size, which may have limited the detection of significant associations. The relationship between surgical complications and hospital admission duration is also an essential topic of discussion. While patients with intraoperative or postoperative complications and arrhythmias tended to have more extended hospital stays, statistical analysis did not reveal a significant association. This could be due to a variety of factors, including the complexity of each individual's recovery process and the hospital's discharge criteria, which may not be based solely on arrhythmia resolution.
Recent advancements have significantly refined the management of post-LVA arrhythmias. Beyond traditional resection, the integration of preoperative/intraoperative 3D Electroanatomic Mapping (EAM) enables more precise identification of arrhythmogenic substrate/foci and can guide targeted ablation strategies during hybrid or open surgical procedures [41-43]. Furthermore, combining LVA resection/ surgical ventricular reconstruction (SVR) with adjunctive surgical VT procedures (eg, cryoablation/endocardial substrate modification) has been reported to improve long-term VT control, supporting the concept that geometric reconstruction alone may not fully eliminate re-entrant circuits [30, 42, 44, 45]. In terms of prevention, early perioperative beta-blocker optimization remains a key evidence-based strategy for reducing post-cardiac-surgery tachyarrhythmias, and perioperative amiodarone prophylaxis has RCT-level evidence for reducing postoperative tachyarrhythmias, with reported reduction in sustained ventricular tachyarrhythmias as well [46, 47]. However, for patients deemed high-risk (eg, persistent complex ventricular ectopy/NSVT, sustained VT, or markedly reduced LVEF after optimization), timely ICD implantation is a guideline-supported standard of care for prevention of sudden cardiac death in eligible patients (eg, ischemic cardiomyopathy with LVEF ≤35% despite optimal therapy, per contemporary guideline criteria) [22]. Finally, improved imaging, particularly Late Gadolinium Enhancement (LGE) cardiac MRI for scar and border-zone characterization, is increasingly central for arrhythmic risk stratification and may inform downstream ICD selection and/or VT ablation planning [48-50].
5. LIMITATIONS AND RECOMMENDATIONS
While this study provides valuable insights into postoperative outcomes following ventricular aneurysm surgery, several limitations warrant consideration. Primarily, the relatively small sample size constrained the statistical power of the study, precluding the feasibility of multivariate analysis to adjust for potential confounders. Consequently, our findings regarding gender-based differences and clinical correlations should be interpreted as exploratory and hypothesis-generating rather than definitive. Additionally, the single-center nature of the research may limit the generalizability of the results to broader clinical settings. Future research should focus on larger, multicenter prospective trials to validate these findings and refine risk stratification models. Long-term studies assessing arrhythmia recurrence, survival rates, and quality of life post-LVA resection are necessary to further establish the durability of surgical benefits and the impact of postoperative complications. Furthermore, integrating advanced imaging and electrophysiological mapping could offer deeper insights into the underlying mechanisms of post-surgical arrhythmias.
6. CLINICAL IMPLICATIONS
Patients with dyslipidemia are at a higher risk of preoperative and postoperative arrhythmias. Aggressive management of lipid profiles, including statin therapy, should be prioritized before surgery to reduce arrhythmic risk. Male patients exhibited a higher incidence of arrhythmias post-LVA resection. Clinicians should be vigilant in monitoring male patients for arrhythmias and consider prophylactic antiarrhythmic therapy or closer postoperative surveillance. Given the high incidence of postoperative arrhythmias (20.7%), continuous ECG monitoring in the immediate postoperative period is essential. Early detection of ventricular arrhythmias (e.g., PVCs, VT) and AF can guide timely interventions such as antiarrhythmic medications, catheter ablation, or ICD placement. Low cardiac output syndrome and cardiac tamponade were the leading causes of in-hospital mortality. Therefore, hemodynamic monitoring and early use of inotropic support or mechanical circulatory assistance should be considered in high-risk patients. Cardiac tamponade contributed to mortality in this cohort. Meticulous surgical hemostasis and postoperative pericardial drainage protocols should be emphasized to prevent this complication.
The significant improvement in LVEF (from 33.4% to 42.9% at two years) underscores the benefits of LVA resection in restoring cardiac function. However, the persistence of arrhythmias suggests that surgical intervention alone may not fully address electrical instability. Long-term follow-up with echocardiography and arrhythmia monitoring is recommended. Despite improved LVEF, patients remain at risk for heart failure. Guideline-Directed Medical Therapy (GDMT) for heart failure, including beta-blockers, ACE inhibitors, and aldosterone antagonists, should be optimized postoperatively. Dyslipidemia was strongly associated with abnormal ECG findings and severe arrhythmias. Postoperative lipid-lowering therapy should be intensified, and lifestyle modifications should be encouraged to reduce arrhythmic risk. Hypertension and diabetes were prevalent in the cohort. Tight control of blood pressure and blood glucose levels may reduce the risk of arrhythmias and improve overall outcomes. The study highlights the role of surgical manipulation in triggering arrhythmias. Surgeons should aim to minimize myocardial trauma and ischemia-reperfusion injury during LVA resection. Patients with intraoperative or postoperative complications had more extended hospital stays. Implementing Enhanced Recovery After Surgery (ERAS) protocols may reduce complications and shorten hospitalization. While no significant association was found between age and arrhythmias, older patients may still be at higher risk due to comorbidities. Individualized risk assessment and tailored postoperative care are essential. Although no significant link was found between pulmonary disease and arrhythmias, patients with COPD or other pulmonary conditions should receive optimized respiratory care to prevent postoperative pulmonary complications.
CONCLUSION
In conclusion, this study underscores that while ventricular aneurysmectomy significantly improves left ventricular systolic function, it does not eliminate the risk of postoperative arrhythmias. Additionally, dyslipidemia emerged as a critical clinical marker, strongly associated with pathological ECG changes and complex arrhythmias. Although the relatively small sample size necessitates an exploratory interpretation of these results, the data suggest that surgical success should be measured not only by hemodynamic recovery but also by long-term electrical stability. Future management strategies should prioritize aggressive lipid-lowering therapy and vigilant postoperative monitoring, particularly in high-risk patients, to mitigate the burden of arrhythmic events and improve overall survival.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: Y.M., K.GH., A.M.: Study conception and design; A.B., H.H.: Analysis and interpretation of results; A.R.A., N.M., M.H.M., A.A.SH., A.A.T.: Data curation; R.R.: Writing - reviewing and editing; K.GH.: Conceptualization. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| LVA | = Left Ventricular Aneurysm |
| MI | = Myocardial Infarction |
| LVEF | = Left Ventricular Ejection Fraction |
| ECG | = Electrocardiography |
| ICU | = Intensive Care Unit |
| VT | = Ventricular Tachycardia |
| PVC | = Premature Ventricular Contraction |
| AF | = Atrial Fibrillation |
| LBBB | = Left Bundle Branch Block |
| CABG | = Coronary Artery Bypass Grafting |
| NSVT | = Non-Sustained Ventricular Tachycardia |
| EAM | = Electroanatomic Mapping |
| SVR | = Surgical Ventricular Reconstruction |
| GDMT | = Guideline-Directed Medical Therapy |
| LDL-C | = Low-Density Lipoprotein Cholesterol |
| RCT | = Randomized Controlled Trial |
| LGE-MRI | = Late Gadolinium Enhancement Cardiac Magnetic Resonance Imaging |
| HIS | = Hospital Information System |
| BMI | = Body Mass Index |
| ARBs | = Angiotensin II Receptor Blockers |
| CASS | = Coronary Artery Surgery Study criteria |
| SPSS | = Statistical Package for the Social Sciences |
| IQR | = Interquartile Range |
| SD | = Standard Deviation |
| 3D EAM | = Three-Dimensional Electroanatomic Mapping |
| VF | = Ventricular Fibrillation |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was conducted in accordance with ethical standards and was approved by the University's Research Council under project number A-10-116-7 on August 19. 2020. Ethical approval was obtained from the Ethics Committee of Semnan University of Medical Sciences (Approval code: IR.SEMUMS.REC.1399.290, dated January 26, 2021).
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
The ethics committee waived the need for informed consent for the use of retrospective data.
AVAILABILITY OF DATA AND MATERIALS
The data generated and analyzed during this study will be made available upon request to the corresponding author, K.GH.
ACKNOWLEDGEMENTS
The authors would like to extend their sincere gratitude to Mashhad University of Medical Sciences and Semnan University of Medical Sciences for their support and collaboration in this research. They also sincerely appreciate the efforts of all individuals who contributed to the data collection, patient care, and manuscript preparation. Their dedication and cooperation were invaluable to the successful completion of this study.


